Glaucoma Medications: When good drugs do bad things
IOP-lowering agents, like all other medicines, have the potential to produce both clinical benefits and harmful effects.
By Bruce E. Onofrey, OD, RPh
Release Date: July 15, 2019
Expiration Date: July 15, 2022
Estimated Time to Complete Activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine (PIM) and Review Education Group
 |
Educational Objectives: After completing this activity, the participant should be better able to:
- Identify the side effects, ocular and systemic, of all types of glaucoma medications, new and old.
- Determine treatment options based on factors of efficacy, safety, risk and compliance.
- Evaluate the benefits and risks of additive/adjunctive medication.
- Incorporate strategies to prevent, reduce, or reverse medication side effects in glaucoma patients.
Target Audience: This activity is intended for optometrists engaged in the care of patients with glaucoma.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and Review Education Group. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education, and the American Nurses Credentialing Center, to provide continuing education for the healthcare team. Postgraduate Institute for Medicine is accredited by COPE to provide continuing education to optometrists.
Faculty/Editorial Board: Bruce Onofrey, OD, RPh, University of Houston.
Credit Statement: This course is COPE approved for 2 hours of CE credit. Course ID is 63132-PH. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements:
Dr. Onofrey has nothing to disclose.
Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The Review Education Group planners, managers and editorial staff have nothing to disclose.
In the late 1800s, the medical management of glaucoma was simple. The one and only choice of treatment was topical pilocarpine, a miracle drug at the time. Over the many decades of its use, patients have certainly enjoyed pilocarpine’s clinical benefits but have also suffered its side effects.
Today, our choices of glaucoma medications are significantly greater. We have multiple drugs that work well as monotherapy, along with many that we can add to improve the clinical outcome with combination drug approaches. Unfortunately—perhaps inevitably—these drugs carry side effects of their own.
This article discusses the concept of rational drug selection and specifically identifies the side effects and adverse events of medications designed to treat the various forms of glaucoma (Table 1).
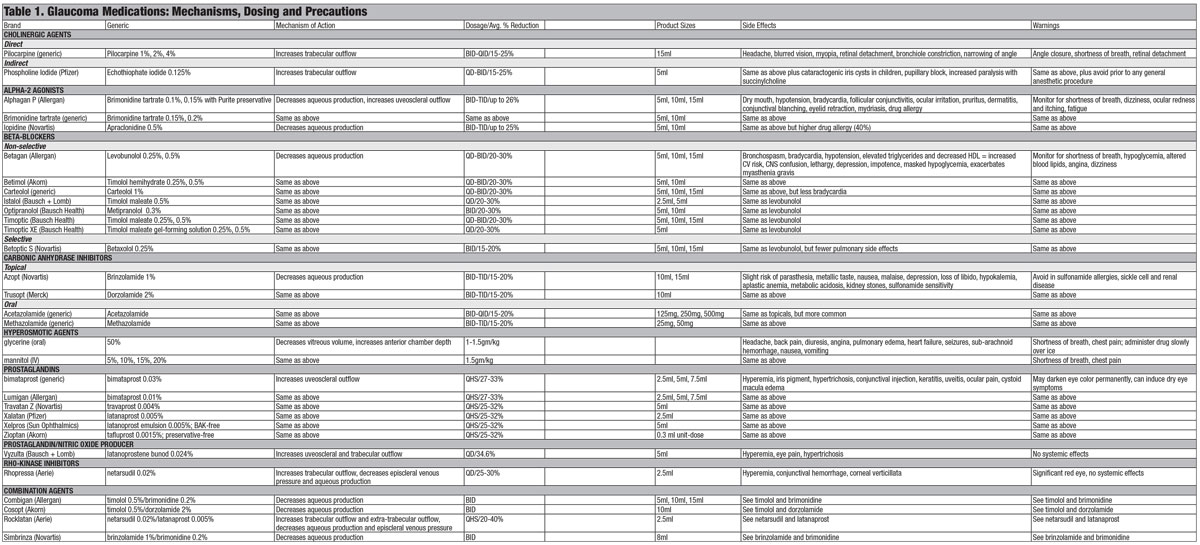 |
| Click table to enlarge, or click here to download a PDF version. |
First, let’s review some basic pharmacology concepts and terms. The pharmacology of any drug includes the drug’s mechanism of action, indications, side effects, adverse events, dosages, dosage forms and warnings.1 A brief definition of each of these terms is warranted:
Pharmacokinetics: The movement of medicine into, through and out of the body—the progression of its absorption, bioavailability, distribution, metabolism and excretion.
Mechanism of action: How the drug interacts with the patient’s physiology and alters it to cure or control a certain disease state.
Indication: The condition for which the US Food and Drug Administration has approved the drug to be safe and effective to treat.
Contraindication: Pre-existing condition in which a drug is not safe or effective in a given individual.
Side effects: Undesirable effects of a drug that are predictable by the drug’s mechanism of action.
Adverse events: Undesirable effects of a drug that are not predictable by the drug’s mechanism of action. They can be dose-dependent (toxic drug effects) or non-dose-dependent (allergic reactions).
Dosages: The calculated amount of safe and effective drug that can be administered to a patient. This calculation can be based on age, weight and renal and hepatic function.
Dosage forms: These include topical solutions, suspensions, emulsions and ointments as well as systemic tablets, suppositories, capsules and injectable forms.
Warnings: Patient counseling that includes significant symptoms that may indicate toxic undesirable effects of a drug. Symptoms can be annoying but non-life-threatening and tolerable, or may indicate a potential life-threatening condition that requires the immediate discontinuation of the drug.
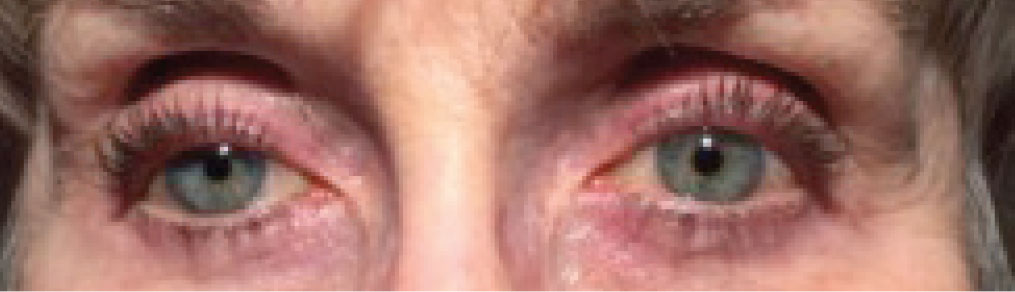 |
| This patient has prostaglandin-associated periorbitopathy with deepened sulci bilaterally due to periorbital fat atrophy and ptosis. After topical treatment with a PGA for more than 10 years, she also has a relative endophthalmos with a sunken globe appearance. Photo: Leslie O’Dell, OD. Click image to enlarge. |
Rational Prescribing
In pharmacology there is a singular truth: drugs have the potential to produce clinical benefits and the potential to produce harm, otherwise known as the risk/benefit ratio.1 It’s up to the clinician to select the right drug for the right disease for the right patient:
1. For any disease, the clinician should choose the most effective drug or drug combination that provides the best chance to control or cure the condition (i.e., benefit).
2. The clinician, after performing a thorough medical interview with the patient—including their medical, social, surgical and ocular histories—can then determine the best drug/dose to treat the given condition while attempting to minimize adverse events/side effects (i.e., risk).
Weighing benefits vs. risks requires a thorough knowledge of the pharmacology of the drug, the pathophysiology of the condition and the medical history of the patient.1
Special Populations
Within the general population, certain patients have pre-existing conditions that could potentially predispose them to increased side effects or adverse events due to their condition (Table 2).
Treatment Goals
The goal of the pharmaceutical management of glaucoma is to select a drug or group of drugs that safely lowers the patient’s intraocular pressure (IOP) while minimizing clinically significant undesirable effects. The concept of combining two or more drugs that produce an effect that is greater than the sum of their separate effects is known as synergy. It is based on the selection of drugs that do the same thing (i.e., lower IOP) but use different mechanisms of action. By this process, one drug enhances instead of interferes with or inhibits the other drug(s).
Additionally, we now know that glaucoma risk is not simply based on IOP—alterations in cardiovascular dynamics (which include blood pressure and pulse), diurnal fluctuations of IOP and use of concurrent systemic cardiovascular agents all can limit the drugs that a patient can safely use to treat their form of glaucoma.2
Currently, the only way to lower the risk of glaucoma progression is by reducing IOP. Depending on the selected agent, this is accomplished by: (1) decreasing aqueous production; (2) increasing trabecular outflow; (3) increasing extra-trabecular outflow; or (4) reducing episcleral venous pressure.
We select medications that maximize each individual factor to increase efficacy, and we avoid drugs that, in each patient, may increase their risk of significant, undesirable clinical effects. Most importantly, we attempt to maximize compliance of therapy by minimizing cost and side effects and maximizing convenience.
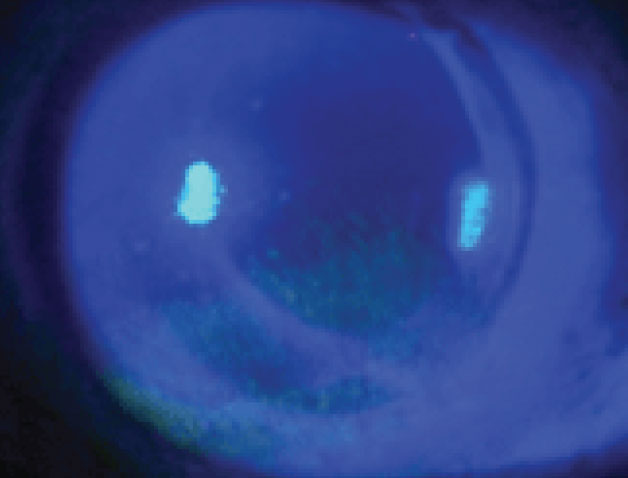 |
| Superficial punctate keratitis is a possible finding associated with CAIs. Photo: Walt Whitley, OD, MBA. Click image to enlarge. |
Aqueous Physiology
All current glaucoma drugs manage the disease by some alteration of aqueous dynamics.
The aqueous is produced by the ciliary body via ultra-filtration, secretion and diffusion. It is formed by the ciliary processes, each of which is composed of a double layer of epithelium. Within each ciliary process, the apical surface of the outer pigmented layer faces the apical surface of the inner non-pigmented layer, and they’re joined by tight junctions. Sandwiched between these two layers is stromal tissue, along with a system of fenestrated capillaries. The inner non-pigmented epithelial cells produce the aqueous.
The production of aqueous humor is controlled by factors that include the enzyme carbonic anhydrase along with blood flow to the ciliary processes.3 This is an important component of the blood-aqueous barrier. The autonomic nervous system, in controlling the afferent blood flow (to the process) and efferent blood flow (from the process), has a significant impact on aqueous production, which is normally approximately 2µL/minute.3 About 90% of aqueous outflow is through the trabecular meshwork, with the remaining 10% through extra-trabecular outflow mechanisms.4
Episcleral venous pressure is now recognized as a new parameter that can be pharmacologically manipulated. It is inversely related to trabecular outflow, and lowering episcleral venous pressure enhances trabecular outflow (assuming the angle is open).5
Glaucoma Drugs
Here is a look at each class of medication, including the ocular and systemic effects:
Cholinergics. The discovery of the autonomic nervous system, neurotransmitters and receptor physiology led to the development of many of the modern drugs used today. Because these receptors are located on every organ of the body, this class of drugs allows for the significant manipulation—and as a result, significant systemic side effects—that are associated with the parasympathetic component of the autonomic nervous system.
Cholinergic drugs mimic the effects of the neurotransmitter acetylcholine. When released from the postganglionic terminals, acetylcholine binds to parasympathetic receptors producing a local response. The enzyme acetylcholinesterase then neutralizes the neurotransmitter and stops the parasympathetic activity.
The physiology of this process is important in predicting the side effects produced by cholinergic glaucoma drugs. These drugs fall into two major categories: direct- and indirect-acting cholinergic agents. The direct-acting agents mimic acetylcholine and directly stimulate the parasympathetic receptors. Indirect (or mixed-acting) agents block the enzyme acetylcholinesterase that deactivates acetylcholine. The indirect agents have the same side effects as the direct-acting agents.6
The mechanism of action of these drugs and their side effects are intimately related.7 They reduce IOP by physically producing contraction of the ciliary body, which increases trabecular outflow. Along with increased trabecular outflow, patients may get other undesirable effects.
Potential ocular side effects:
- Stinging, burning and redness
- Headache/brow ache
- Miosis
- Increased accommodation
- Ciliary spasm
- Induced myopia
- Reduced acuity
- Posterior synechiae
- Retinal detachment (rare)
Contraction of the ciliary body is responsible for many of these undesirable effects.7 Miosis can be problematic in individuals with lens opacities. Certainly, in the presence of intraocular inflammation, miosis increases the risk of posterior synechia and the development of pupil block. For these reasons, cholinergics are contraindicated in patients with intraocular inflammation or angle-closure glaucoma.
The worst of the cholinergics are the indirect-acting agents with long half-lives, such as the phosphorylating agent Phospholine Iodide (echothiophate iodide, Pfizer).7 A major concern in the use of these agents is a potential interaction with succinylcholine, a drug used to paralyze muscles during general surgery. Use of these agents must be discontinued three weeks prior to the use of general anesthesia to prevent a risk of increased apnea in the operated patient.8
Table 2. Special Populations
|
Potential systemic side effects:
- Bronchospasm in asthmatics
- Nausea, vomiting and diarrhea associated with increased GI motility
- Bradycardia via stimulation of the vagus nerve (CN X)
- Salivation and sweating via stimulation of exocrine glands
- Central nervous system depression
Alpha-2 agonists. While alpha-1 receptors tend to produce vasoconstriction and raise blood pressure, alpha-2 receptors tend to inhibit alpha-1 activity. As a result, stimulation of alpha-2 receptors leads to decreased blood pressure, sedation and aqueous production reduction.9
The original alpha-2 agonist was clonidine, which is most commonly used systemically to control blood pressure.10 The current glaucoma medications apraclonidine and brimonidine are derivatives of clonidine. Again, these drugs do not affect aqueous outflow, but do reduce aqueous production. The side effects of these drugs are closely tied to clonidine and its alpha-2 activity and partial alpha-1 activity.11
Potential ocular side effects:
- Conjunctival blanching
- Lid retraction
- Mydriasis/miosis
- Significant incidence of ocular allergy (apraclonidine 36% within 90 days and brimonidine 10% within one year)
- Follicular conjunctivitis
Potential systemic side effects:
- Dry mouth
- Sedation
- Hypotension
- Reduced optic nerve perfusion (due to drop in blood pressure)
Beta-blockers. Drugs that block sympathetic beta-1 and beta-2 receptors were discovered in 1958. The original non-selective (B1 and B2) blocker is propranolol.12 It was designed to treat elevated blood pressure, cardiac arrhythmias and angina. When researchers discovered that this drug could also lower IOP by reducing aqueous production, the search for a useful topical agent began. Propranolol proved to be unacceptable because of its corneal side effects, including profound dry eye, corneal anesthesia and subconjunctival scarring.
Eventually, the non-selective beta-blocker timolol revolutionized glaucoma management in 1978.13 Although ocular side effects are generally mild, the non-selective beta-blockers are notorious for cardiac and pulmonary side effects. The identification of beta-1 selective agents improved the safety of this group in those with chronic obstructive pulmonary disease (COPD) and asthma.13
Potential ocular side effects:
- Dry eye
- Ptosis
- Conjunctival hyperemia
- Corneal anesthesia
- Blurred vision
- Cicatricial ocular pemphigoid (timolol)
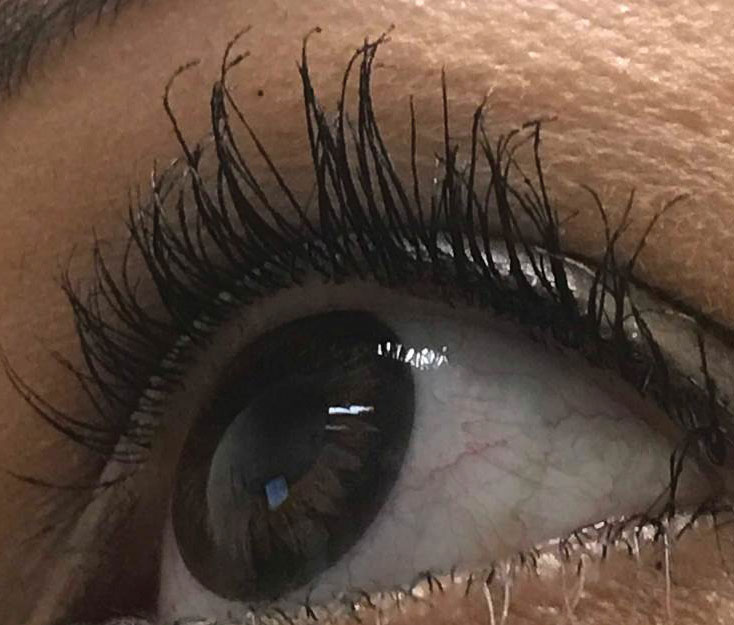 |
| Trichomegaly resulting from prolonged use of prostaglandins. Photo: Bisant A. Labib, OD. Click image to enlarge. |
Potential systemic side effects:
- Cardiovascular
- Bradycardia (less with carteolol)
- Systemic hypotension
- Arrhythmia
- Angina
- Heart attack
- Heart block
- Death (anaphylaxis reversal)
- Pulmonary (less with betaxolol)
- Bronchospasm
- Wheezing
- Dyspnea
- Cough
- Central nervous system
- Depression
- Sedation
- Fatigue
- Confusion
- Inhibits SSRIs
- Memory loss
- Metabolic side effects
- Increased cardiac risk
- Decreased high-density lipoproteins
- Elevated serum triglycerides
- Increased risk of coronary heart disease
- Diabetic hypoglycemia
Take note that topical beta-blockers are able to reach significant blood levels. Eighty percent of the drug leaves the eye via the nasolacrimal system, with a large portion of that being directly absorbed through the nasolacrimal mucosa. When a drug is absorbed by this pathway it avoids the first pass effect—that is, it avoids being initially metabolized by the liver.14 This produces an effect similar to directly injecting the drug into the bloodstream.
Due to this first pass effect, topical administration of beta-blockers can lead to autonomic, life-threatening side effects. The most significant cardiopulmonary side effects are bronchiolar constriction and bradycardia. Beta-2 blockade of the lungs produces life-threatening bronchiolar constriction in those with COPD and asthma, and should be avoided in these populations. Beta-1 blockade produces bradycardia, hypotension and a drop in systemic perfusion that can lead to end organ ischemia. Patients who are treated with these agents can have their pulmonary function evaluated very simply with an in-office peak flow test and should have their blood pressure and heart rate checked at each visit.
Beside the cardiovascular and pulmonary side effects seen with beta-blockers, certain individuals with diabetes are at special risk. People with either Type 1 or 2 diabetes can use beta-blockers as long as they aren’t prone to significant hypoglycemic events. When blood glucose drops, the body releases epinephrine to stimulate the liver to convert glycogen to glucose (gluconeogenesis). The symptoms of hypoglycemia produced by high levels of epinephrine produce symptoms (tremor, tachycardia, sweating, etc.) that warn the person with diabetes of the risk of going into ketoacidosis from hypoglycemia, which should lead the individual to consume glucose and alter their therapy to avoid coma and death. However, the use of beta-blockers can reduce these warning signs of hypoglycemia, so they should be avoided in any person with diabetes who is prone to hypoglycemia.15
Another special situation is when any patient has a significant, life-threatening allergic reaction (anaphylaxis) that requires self-administration of epinephrine (Epipen, Mylan). Beta-blocker use reduces effectiveness of the life-saving epinephrine during an anaphylactic event.15
Patients taking antidepressant therapy can expect a significant reduction in the effect of their medication with highly lipid soluble drugs (timolol) and should avoid using it.15
Patients taking systemic beta-blockers should avoid the topical form. The oral agent reduces the efficacy of the topical agent, and the sum of the two increases the risk of side effects. So, the two drugs produce little therapeutic benefit but one sees an increase in drug toxicity.16
Patients with heart block should definitely avoid these drugs. They can lead to an increase in heart block, including a fatal arrhythmia.17
Patients taking calcium channel blockers are particularly prone to bradycardia and/or significant hypertension if used with topical beta-blockers.18
Carbonic anhydrase inhibitors (CAIs). The CAIs acetazolamide and methazolamide have been used for decades to manage significant elevation of IOP produced by secondary forms of open-angle glaucoma such as uveitic and hemolytic glaucoma, as well as primary angle-closure glaucoma and, in the past, poorly controlled primary open-angle glaucoma. The systemic side effects of these drugs are significant and certainly limit their use today in the management of primary open-angle glaucoma. The development of topical CAIs in the late 1990s have largely replaced the use of oral agents except in extreme presentations of acute rise in IOP.19
As their name implies, CAIs inhibit the enzyme carbonic anhydrase, whose responsibility is the production of bicarbonate ion, important in stabilizing the body’s pH, specifically in the kidney. Oral use of the drug must block a substantial amount of carbonic anhydrase activity before it results in a significant reduction in aqueous production. This marked reduction in carbonic anhydrase activity is responsible for the systemic side effects of these drugs and why most primary open-angle glaucoma is managed with the topical form of the drug available as dorzolamide and brinzolamide.19
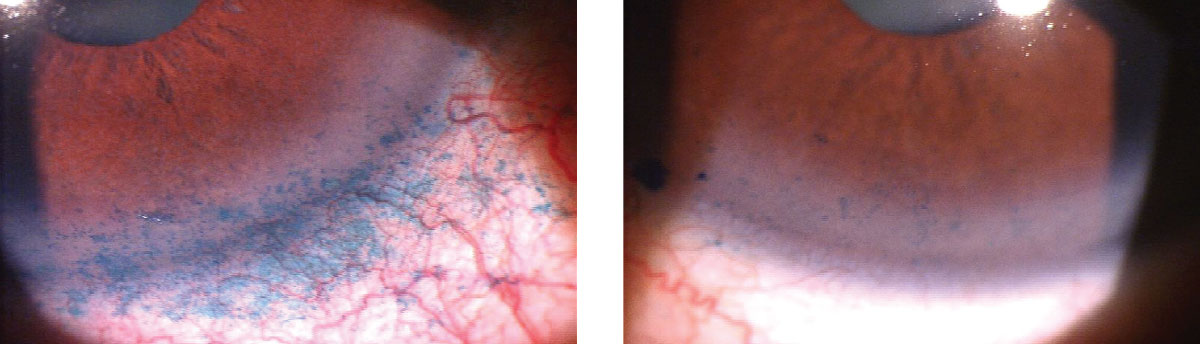 |
| Lissamine green staining of the conjunctiva and cornea identifies BAK toxicity from a prostaglandin. This patient was switched to a preservative-free prostaglandin analog. His signs and symptoms of dry eye improved over the next three months. Photos: Paul C. Ajamian, OD. Click image to enlarge. |
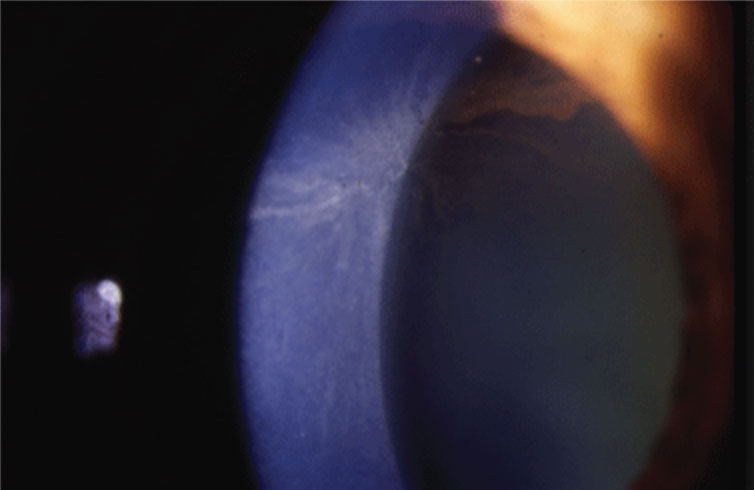 |
| Corneal verticillata may be a common side effect of rho-kinase inhibitor use. Photo: Jay S. Pepose, MD, PhD. Click image to enlarge. |
Potential ocular side effects (topical drugs):
- Burning, stinging and tearing
- Corneal edema (altered endothelial cell function)
- Potential sulfonamide sensitivity (low risk)
- Bitter aftertaste (dysgeusia)
- Allergic reactions of the conjunctiva
- Superficial punctate keratopathy
Potential systemic side effects (oral drugs):
- Metabolic acidosis
- Sickle cell crisis (increased risk of sickling associated with metabolic acidosis)
- Stevens-Johnson syndrome
- Sulfonamide reaction
- Aplastic anemia
- Dysgeusia
- Anorexia
- Weight loss
- Paresthesia
- Malaise
- Fatigue
- Depression
- Hypokalemia
Prostaglandins. The development and use of topical prostaglandin analogs has revolutionized the management of glaucoma. Their high efficacy and relative lack of significant systemic side effects have made them the drug of choice for treating ocular hypertension and glaucoma. Their unique mechanism of action reduces IOP through increased uveoscleral outflow.
Because prostaglandins are highly effective and dosed only once a day, they’re an ideal option for a single-drug treatment strategy. When a stronger effect is required, combining a prostaglandin with any other topical hypotensive agent(s) results in a synergistic and potent way to maximize the reduction of IOP.
The main side effects of prostaglandins are ocular.20
Potential ocular side effects:
- Hyperemia
- Hypertrichosis (increased eyelash growth)
- Darkening of the lids and periocular skin
- Permanent change in iris coloration (darkening)
- Cystoid macular edema
Potential systemic side effects:
- Headache
Nitric oxide generators. The recently approved Vyzulta (latanoprostene bunod, Bausch + Lomb) is a prostaglandin with the addition of bunod. The bunod portion is a nitric oxide (NO) donator that enhances trabecular outflow in addition to the increase in extra-trabecular outflow from the prostaglandin portion of the drug.21
The addition of bunod does not add any additional ocular or systemic side effects.
Rho kinase inhibitors. The most recently approved addition to our collection of topical IOP-reducing agents is the rho-kinase inhibiting drug netarsudil, marketed as Rhopressa and as the netarsudil/latanoprost combination drug Rocklatan (both from Aerie Pharmaceuticals).
Netarsudil’s mechanism of action and side effect profile are unique to this singular class of anti-glaucoma agents—it’s the only drug that has a triple mechanism of action. Netarsudil is able to increase trabecular outflow, reduce aqueous production and decrease episcleral venous pressure.
As unique as the drug is in its mechanism of action, it also displays some significant and unique ocular side effects.22
Potential ocular side effects:
- Conjunctival hyperemia
- Corneal verticillata
- Eye pain
- Subconjunctival hemorrhage
- Blurred vision
- Tearing
- Reduced acuity (5% to 10% reported)
Potential systemic side effects:
- None
The main responsibility of primary care providers, like optometrists, is to continuously monitor their patients for adverse events. Some of these adverse events are exacerbated by medications that we prescribe or by those that others prescribe. Continuously updating patients’ medical and drug histories, and inquiring about any new symptoms, allows us to detect and prevent the undesirable and sometimes disastrous side effects that certain drugs can produce.
All treatments have both risks and benefits. Choosing the proper treatment requires a thorough knowledge of the patient’s histories. Anticipating side effects, excluding patients with allergies or other contraindications, and educating patients on potential adverse events helps to avoid preventable complications.
Dr. Onofrey is a clinical professor and the executive director of continuing education programs at the University of Houston.
|
1. Hodgson E, Levi PE eds. Introduction to Biochemical Toxicology. 2nd ed. Norwalk, CT: Appleton and Lange; 1994. 2. Hoffman BB, Lefkowitz RJ. Alpha-adrenergic receptor subtypes. N Engl J Med. 1980;302(25):1390-6. 3. Kim YW, Park KH. Exogenous influences on intraocular pressure. Br J Ophthalmol. 2019 Mar 25. [Epub ahead of print]. 4. Onofrey BE, Holdeman NR, Skorin L. Ocular Therapeutics Handbook. 4th ed. Philadelphia: Wolters Kluwer; 2019. 5. Kaufman PL. Aqueous Humor Dynamics. In: Duane TD, ed. Clinical Ophthalmology. Philadelphia: Harper and Row; 1985: 1-24. 6. Leopold IH. The use and side-effects of cholinergic agents in the management of intraocular pressure. In: Glaucoma: Applied Pharmacology. Orlando: Grune and Stratton; 1984: 357-93. 7. Kini MM, Dahl AA, Roberts CR, et al. Echothiophate, pilocarpine, and open-angle glaucoma. Arch Ophthalmol. 1973 Mar;89(3):190-2. 8. Bito LZ, Hyslop K, Hyndman J. Anti-parasympathetic effect of cholinesterase inhibitor treatment. J Pharmacol Exp Ther. 1967;157:159-169. 9. Alquist RP. A study of adrenotropic receptors. Am J Physiol. 1948;153:586-599. 10. Krieglstein GK, Langham ME, Leydhecker W. The peripheral and central neural actions of clonidine in normal and glaucomatous eyes. Invest Ophthalmol Vis Sci. 1978;17(2):149-58. 11. Robin AL. Short-term effects of unilateral 1% apraclonidine therapy. Arch Ophthalmol. 1988;106(7):912-5. 12. Phillips CI, Howitt G, Rowlands DJ. Propranolol as ocular hypotensive agent. Br J Ophthalmol. 1967;51(4):222-6. 13. Offner S, Smith TJ. Betaxolol in chronic obstructive pulmonary disease. J Ocul Pharmacol. 1987;3(2):171-6. 14. Atkins JM, Pugh BR Jr, Timewell RM. Cardiovascular effects of topical beta-blockers during exercise. Am J Ophthalmol. 1985;99(2):173-5. 15. Schmidt D, Schmetterer L, Garhofer G, et al. Pharmacotherapy of glaucoma. J Ocul Pharm Ther. 2015;31(2):63-77. 16. Gulea C, Zakeri R, Quint JK. Effect of beta-blocker therapy on clinical outcomes, safety, health-related quality of life and functional capacity in patients with chronic obstructive pulmonary disease (COPD): a protocol for a systematic literature review and meta-analysis with multiple treatment comparison. BMJ Open. 2018 Nov 13;8(11):e024736. 17. Nelson WL, Fraunfelder FT, Sills JM, et al. Adverse respiratory and cardiovascular events attributed to timolol ophthalmic solution, 1978-1985. Am J Ophthalmol. 1986 Nov 15;102(5):606-11. 18. Pringle SD, MacEwen CJ. Severe bradycardia due to interaction of timolol eye drops and verapamil. Br Med J (Clin Res Ed). 1987;294(6565):155-6. 19. Hoffmanová I, Sánchez D. Metabolic acidosis and anaemia associated with dorzolamide in a patient with impaired renal function. Br J Clin Pharmacol. 2018;84(4):796-799. 20. Kim HY, Lee SK, Lee JH, et al. A case of latanoprost-induced diffuse facial skin hyperpigmentation. J Cosmet Dermatol. March 28, 2019. [Epub ahead of print]. 21. Fingeret M, Gaddie IB, Bloomenstein M. Latanoprostene bunod ophthalmic solution 0.024%: a new treatment option for open-angle glaucoma and ocular hypertension. Clin Exp Optom. January 7, 2019. [Epub ahead of print]. 22. Kahook MY, Serle JB, Mah FS, et al. Long-term safety and ocular hypotensive efficacy evaluation of netarsudil ophthalmic solution: Rho Kinase Elevated IOP Treatment Trial (ROCKET-2). Am J Ophthalmol. 2019;200:130-37. |
