9th Annual Retina ReportFollow the links below to read other articles from annual update on retina: Busted Barriers: Triaging Retinal Hemorrhages |
In 2017, the Optometric Retina Society (ORS) chose to honor the legacy of Larry Alexander, OD, by launching the Larry Alexander Case Report Contest for optometric residents. The contest was made possible by support from Optovue. Submissions were reviewed independently by the ORS board. The case below is this year’s winner.
A patient who presents to the eye clinic with an acute red eye, decreased vision and concurrent eye pain can represent a vast array of pathologies. Our diagnostic challenge as clinicians is to distinguish the everyday dry eye syndrome from the rare ophthalmic emergencies that can have sight-threatening complications such as endophthalmitis.
Endophthalmitis is a rapidly progressing purulent inflammation of the aqueous and vitreous that usually stems from an infection.1
This report outlines a rare case of endogenous endophthalmitis, in an immunocompromised male patient, that was secondary to endocarditis with methicillin-resistant Staphylococcus aureus (MRSA) sepsis.
It also details common clinical findings, disease process, microorganism profile and proper treatment strategies for the different forms of endophthalmitis.
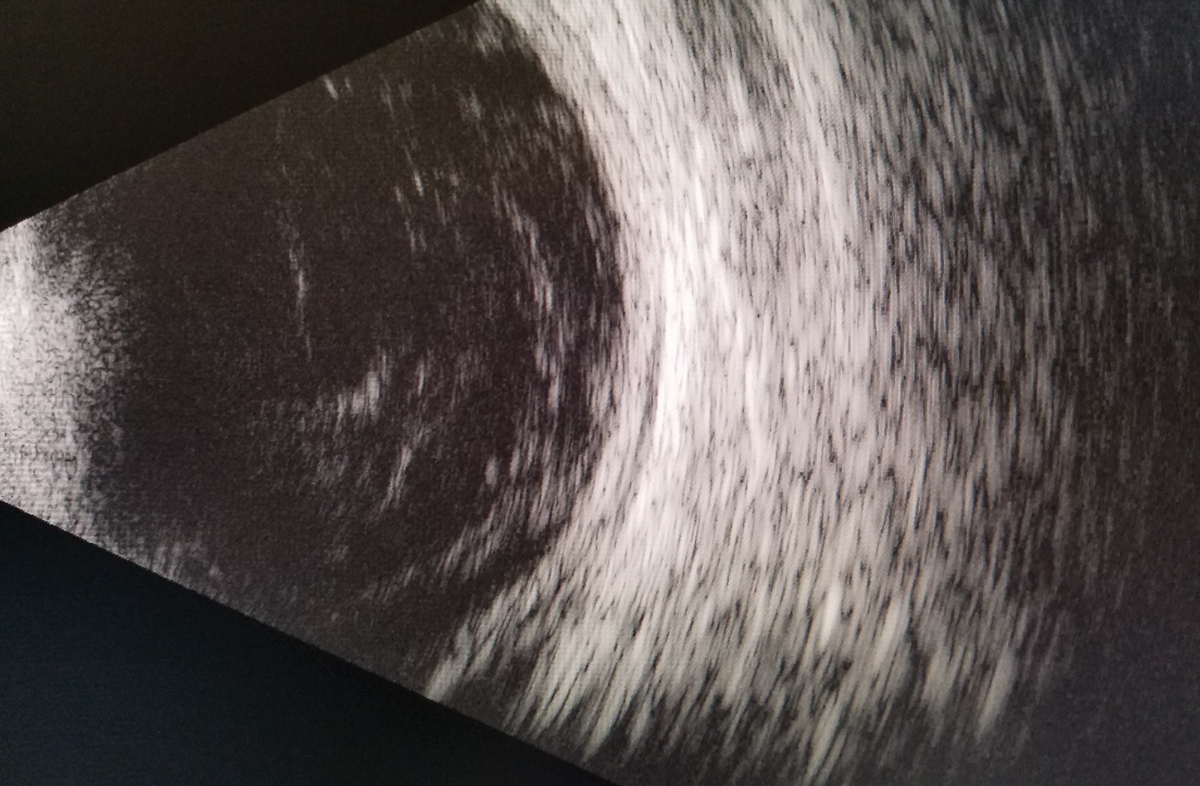  |
| Fig. 1. A B-scan of the right eye reveals vitritis with no retinal detachment or mass. Click image to enlarge. |
Presentation
A 61-year-old Caucasian male presented emergently for examination complaining of sudden vision loss in his right eye over the last two days. He rated his pain as a “five” on a scale of one to 10 and reported seeing “white swiggly lines” that had worsened since onset. He denied other visual symptoms. His last examination five months prior was unremarkable. His ocular history, however, was remarkable for partial lower left excision for basal cell carcinoma and previous alpha-2a interferon treatment for corneal intra-epithelial neoplasm of the right eye. The patient’s medical history was positive for Type II diabetes mellitus and a history of alcohol, cocaine and intravenous drug use. The patient’s family history was non-contributory. He had no known drug allergies and was oriented to time, place and person.
Uncorrected visual acuity (VA) was counting fingers at one foot in his right eye and 20/25-2 in the left. The right pupil was minimally reactive with a trace afferent pupillary defect. The left pupil was round and reactive to light. Extraocular muscles were unrestricted in all gazes without pain or diplopia. Confrontation fields were constricted in all inferior quadrants in the right eye and were full in the left eye. Cover test was orthophoric at distance and near. On red-cap desaturation testing, the patient reported “blue” cap in the right eye, 100% red in the left. Pseudo-isochromatic color plates were attempted but not completed due to poor patient cooperation and fatigue.
Slit lamp biomicroscopy revealed normal external adnexae in both eyes. The left anterior ocular structures were normal unless otherwise stated. The right bulbar conjunctiva had trace diffuse injection. The right cornea was remarkable for grade 1 Descemet’s folds centrally with diffuse microcystic edema. The right anterior chamber had grade 3-4 cells with grade 1+ flare and a 1.5mm hypopyon inferiorly. Both irides were flat and intact. The anterior chamber depth was normal. Goldman applanation tonometry measured 12mm Hg OU at 3:10pm.
The patient was dilated using one drop of 1% mydriacyl and 2.5% phenylephrine OU. Evaluation of the posterior segment with a 90D lens revealed grade 2+ nuclear sclerosis of the lens of both eyes. The vitreous media of the right eye had a dense vitritis that prevented further posterior views of the retina. The B-scan ultrasound revealed a dense vitritis and an absence of a retinal detachment or mass (Figure 1).
The left eye fundus findings included: clear vitreal media, a healthy optic nerve with a 0.30 H/V cup-to-disc ratio and distinct margins. There were dot hemorrhages with elevated patches of choroidal whitening along the superior and inferior temporal arcades. The macula was flat and even with two Roth spots inferior to the fovea. The peripheral retina was flat without pathology.
We considered differential diagnoses of panuveitis, chorioretinitis (infectious/inflammatory), malignant neoplasms, intraocular foreign bodies (IOFBs) and endogenous endophthalmitis. After a retinal consultation, the patient was diagnosed with endogenous endophthalmitis.
Intravitreal injections of 0.1ml of 2.25mg/0.1ml ceftazidime and 0.1ml of 5ug/ml amphotericin B were performed without complications. Bacterial and fungal blood cultures were ordered along with an infectious disease consultation to determine the primary source of infection. The patient was started on intravenous vancomycin, Zosyn (piperacillin and tazobactam, Pfizer) fluconazole and clindamycin and was scheduled to return to the eye clinic the following morning for re-evaluation.
Day one follow-up. The patient presented without new visual complaints, stable acuity and stable exam findings in both eyes. The patient received an intravitreal injection of 0.1ml of 1mg/0.1ml vancomycin in his right eye. He was also scheduled to undergo pars plana vitrectomy (PPV) in the right eye in four days if no improvement was found at the follow-up in two days. Human immunodeficiency virus (HIV) labs were also ordered by the infectious disease department.
Day three follow-up. The patient returned with significant improvement in his right eye’s vision. The nurse present at the exam revealed the patient had fever spikes over the previous 48 hours. Uncorrected distance VA was 20/200 OD and 20/25-1 OS. The right bulbar conjunctiva still had trace diffuse injection. The right cornea showed trace Descemet’s folds centrally and temporally with no sodium fluorescein staining. The right anterior chamber had grade 3 cells with no flare and no hypopyon.
Posterior exam in his right eye revealed grade 1 vitritis with a flat and even macula and 0.45 H/V cup-to-disc ratio of the optic nerve with distinct margins. His right eye’s posterior pole had scattered intraretinal hemorrhages with scattered white choroidal lesions and Roth spots (Figure 2). The posterior exam of the left eye was stable from the initial visit. The blood cultures returned positive for MRSA, and bloodwork results showed a newly diagnosed HIV positive status. Given improvement in his right eye acuity and decreased inflammatory reaction, the vitrectomy was not performed.
Day five follow-up. The patient reported progressive improvement in the right eye’s vision. Further work-up by the infectious disease department and primary care physician revealed a positive diagnosis for endocarditis with MRSA sepsis. Uncorrected distance VA was 20/100 OD and 20/30 OS. The bulbar conjunctiva was white and quiet in both eyes. The right cornea showed trace non-granulomatous keratic precipitates inferior with no sodium fluorescein staining. The right anterior chamber had grade 3 cells with no flare and no hypopyon. Dilated fundus examination of the right eye revealed grade 1 vitritis with otherwise stable findings to the previous dilated fundus exam. Given improvement but persistent inflammatory reaction in the right eye, the patient was started on one drop prednisolone acetate 1% TID OD.
Day eight follow-up. The patient returned to clinic after another three days with improvement in his right eye’s vision. Uncorrected distance VA was 20/80 OD and 20/25 OS. The bulbar conjunctiva was white and quiet in both eyes. The right cornea showed resolving trace non-granulomatous keratic precipitates inferior with no sodium fluorescein staining. The right anterior chamber had grade 2 cells with no flare and no hypopyon.
Dilated fundus exam in the right eye revealed trace vitritis with resolving intraretinal hemorrhages, white choroidal lesions and Roth spots. The patient continued one drop prednisolone acetate TID OD. The patient was monitored closely with subsequent follow-up. At last exam his best-corrected VA at distance was 20/25+2 OD and 20/20-1 OS, the anterior chamber was deep and quiet with no cells or flare, no vitritis and rare intraretinal hemorrhages with Roth spots. The patient is currently being treated as in-patient by cardiology for endocarditis.
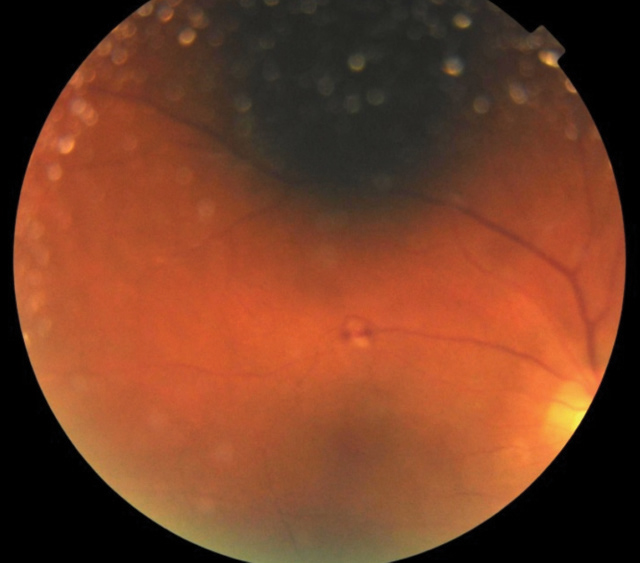 |
| Fig. 2. Presence of Roth spot along superior temporal arcade of the right eye. |
Discussion
Endophthalmitis, a rapidly progressing inflammatory disorder secondary to infection of the vitreal cavity, can present from an exogenous or endogenous source. Exogenous endophthalmitis can arise from acute or chronic postoperative blebitis, trauma or intravitreal injection.
Postoperative endophthalmitis accounts for approximately 70% of exogenous cases.1 Acute cases generally present within the first week. Patients present with severely decreased visual acuity, eye pain, eyelid swelling, mucoid discharge and hyperemia. Examination with biomicroscopy may reveal corneal edema, diffuse bulbar conjunctival injection, hypopyon, dense vitritis and possible retinitis. Of note, approximately 25% of patients will not experience eye pain.1 Acute postoperative cases are most commonly caused by gram-positive, coagulase negative micrococci. Chronic cases, which are differentiated from acute cases by an onset of greater than a six-week duration postoperatively, are commonly caused by slower growing gram-negative bacteria or fungi. One study looked at the microbiologic spectrum of acute postoperative bacterial endophthalmitis and revealed that 94.2% of patients showed a gram-positive bacteria stain while only 6.5% showed a gram-negative bacteria stain.2,3 Of the gram-positive strains, Staphylococcus aureus presented with the highest prevalence (9%), followed by Streptococcus species at 2.2%.2
The presence of a bleb after trabeculectomy surgery presents a continuing risk for infection. Breakdown of the bleb wall allows for microbe infiltration from the tear film and surrounding ocular structures. Early leakage (within three months of surgery) is often secondary to incomplete conjunctival closure or trauma. Investigators suspect that late onset leakage (leakage after three months of surgery) is associated with anti-metabolite use (i.e., mitomycin-C or 5-fluorouracil) during trabeculectomy.4
A 2007 retrospective study found a 15% risk of bleb leakage within five years of surgery associated with the use of anti-metabolite.4 Blebitis presents with sudden onset pain, photophobia, mucoid discharge, hyperemia and possible hypopyon. With its extension into the vitreous, bleb-associated endophthalmitis (BAE) has similar clinical findings to blebitis but with exaggerated signs and symptoms. Due to bleb leakage being the primary source for infection, the most common microbe associated with BAE is Staphylococcus epidermidis, followed closely by the Streptococcus family.6
Even with aggressive early management, BAE cases have a poor visual prognosis. Retrospective studies show 94% of cases result in 20/200 or less visual acuity with approximately 35% resulting in no light perception.6,7
A penetrating globe injury can operate as an access point for opportunistic microbes. The incidence of endophthalmitis after a history of penetrating injury without an IOFB is approximately 12% and jumps to 25% with the presence of an IOFB.8,9 Patients will present with the typical endophthalmitis signs and symptoms: conjunctival injection, purulent discharge, moderate anterior chamber reaction, lid edema, light sensitivity, vitritis and possible hypopyon. Eye pain can range from mild photophobia to severe pain, depending on the type of microbe, making the final diagnosis challenging. Most cases of post-traumatic endophthalmitis are caused by gram-positive bacterial organisms, most often the Bacillus species, especially in the presence of an IOFB or a non-sterile environment.10
While the advent of intravitreal injections, specifically anti-vascular endothelial growth factor (VEGF) injections, has reformed treatment for proliferative retinal diseases such as diabetic retinopathy and macular degeneration, it also presents an avenue for microbial infiltration. A seven-year retrospective study examined 199 cases of endophthalmitis and found that anti-VEGF injections represented 8.5% of all cases.11 Furthermore, analysis of vitreal tap from these cases found the most common culprit to be of the coagulase-negative Staphylococcus family (50% of cases), similar to postoperative endophthalmitis.2,11
Endogenous endophthalmitis accounts for only 2% to 8% of endophthalmitis cases, but is still sight-threatening and must be promptly diagnosed and swiftly managed.12 In endogenous endophthalmitis, the eye is the secondary site of infection, and a full systemic work-up is warranted to find the source of infection. In 44% of cases, no causative source is identified.13 Risk factors include immunocompromised states, recent surgery, pregnancy, endocarditis, intravenous drug use, indwelling catheters and dental procedures.14 The infectious agent travels from its original source via the bloodstream where it then invades the vasculature of the posterior segment and eventually spreads to the vitreous through rupture of a septic embolus.15
Endogenous endophthalmitis signs and symptoms include: red eye, eye pain, reduced visual acuity, photophobia, corneal edema, conjunctival injection, cells and flare, hypopyon and vitritis. Prospective studies from the Americas, Europe, Asia and Australia that performed microbiological analysis found an interesting variation in results. Anterior chamber and vitreal samples that were analyzed through gram staining and culture positivity found gram-positive organisms, most often Staphylococci and Streptococci, to be culprits in the Americas and Europe. In Asia and Europe, gram negative microbes and fungi tended to dominate.16,17
Endogenous endophthalmitis requires rapid management. Necessary ancillary workup should include B-scan ultrasound—especially with the presence of significant media opacification secondary to vitritis—to rule out retinal or choroidal detachments, dislocated lens and the presence of IOFBs. Determining the specific etiology is accomplished with vitreal aspiration or diagnostic PPV, followed by culturing and histology. Vitrectomy has a 92% diagnostic rate of culturing compared with vitreal aspiration (44%).18
Immediate PPV is often performed for several reasons: direct reduction of infectious agents and toxins, clearing of media opacities, better culturing sample and faster clearance of intravitreal antibiotics.19,20 Another emerging technique for identification of endogenous endophthalmitis after procuring vitreous or aqueous samples is real-time polymerase chain reaction (RT-PCR). This procedure has increased sensitivity and specificity compared with culturing, rapid test time (90 minutes) and less worry about contaminated cultures.21,22 However, culturing is still recommended as part of the management plan because RT-PCR does not highlight antibiotic susceptibility.23 Given that endogenous endophthalmitis is a result of systemic infection, it is imperative to perform a blood culture with sterile precautions to find the source.
After obtaining cultures through PPV or vitreal aspiration, empirical intravitreal antibiotic and antifungals are typically initiated. Vancomycin (1.0mg/0.1ml) and ceftazidime (2.25mg/0.1ml) are often the antibiotics of choice for broad-spectrum coverage (gram + and gram -); however, amikacin (400ug/0.1ml) may also be used in penicillin allergic patients.1,23 The use of intravitreal antibiotics may come with complications such as corneal opacification and retinal toxicity. Aminoglycosides, most notably gentamicin, may cause a macular infarct.18,24 In addition to antibiotics, intravitreal amphotericin-B (5-10ug/0.1ml) or voriconazole (100-200ug/0.1ml) are the recommended antifungals of choice due to their broad-spectrum coverage. They carry the same risk of retinal toxicity as antibiotics.18,24
Systemic antibiotics are usually not the treatment of choice for exogenous endophthalmitis due to their poor penetration through the blood-retinal barrier and time needed to be processed systemically. However, in the case of endogenous endophthalmitis, systemic antibiotics and antifungals must be initiated to treat the underlying source of infection and continued for four to six weeks, depending on the patient’s individual health status.
The use of corticosteroids to help reduce intraocular inflammation is controversial. A 2003 study found better visual outcomes with additional treatment with intraocular steroids.25 However, conflicting studies found no improvement in visual outcome with the use of steroids.26 Current corticosteroid use is left to the clinician’s judgment.
This case demonstrates the importance of patient history, awareness of clinical findings and an inter-disciplinary approach for timely intervention of endophthalmitis. Regardless of its source, endophthalmitis can progress rapidly with longstanding ocular and systemic ramifications. However, with proper examination, ancillary testing and treatment the prognosis for patients is favorable.
Dr. Khedekar is finishing a primary care residency at the Bay Pines VAMC in St.Petersburg, FL. He is a graduate of the New England College of Optometry.
Dr. Cordero practices at the Bay Pines VAMC in St.Petersburg, FL, and is acting president of the Pinellas Optometric Association.
1. Forester R. Endophthalmitis Vitrectomy Study Group: Results of the endophthalmitis vitrectomy study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Arch Ophthalmol. 1995;113:1479-96. |

